 1Ġ.057” (1.45 mm) Inner-diameter allows more room to deliver interventional devices.Ġ.066” (1. Hydrophilic coating on the outer diameter reduces friction and enhances deliverability through complex, tortuous anatomyġx1 Braid provides extra back-up support without over-straightening the vesselĠ.001'' Larger I.D. Stiff-yet-flexible stainless steel hypotube shaft provides exceptional pushability and kink resistance when advancing the extension catheterĥ5% Less Frictional Force 1 / Better Deliverability It is strictly forbidden to make copies, whether partial or total and on whichever media without prior approval.Įxtend your reach when treating complex lesions The GuideLiner catheter (Vascular Solutions Inc., Minneapolis, Minnesota, United States) is a coaxial mother and child catheter, mounted on a monorail system, that extends the angioplasty guiding catheter and enables deep intubation of the coronary artery to achieve extra support and improve coaxial alignment. This Website is protected by the laws on copyright and by the relevant international conventions. 
Prior to use, please consult device labeling for prescriptive information and operating instructions. To the extent this site contains information, reference guides and databases intended for use by licensed medical professionals, such materials are not intended to offer professional medical advice. Please note that the following pages are exclusively reserved for health care professionals in countries with applicable health authority product registrations. The ICD-10 HAC Lists for FY 2016 through the current FY year are available in the ICD-10 HAC List section, accessible through the navigation menu at left.For health care professionals in EUROPE excepted those practicing in France as the following pages are intended to all International health care professionals and are not in compliance with the French Advertising law N☂011-2012 dated 29th December 2011 article 34. Other health care professionals should select their country in the top right corner of the website. Iatrogenic Pneumothorax with Venous Catheterization.Deep Vein Thrombosis (DVT)/Pulmonary Embolism (PE) Following Certain Orthopedic Procedures:.Surgical Site Infection Following Cardiac Implantable Electronic Device (CIED).Surgical Site Infection Following Certain Orthopedic Procedures.Laparoscopic Gastric Restrictive Surgery.Surgical Site Infection Following Bariatric Surgery for Obesity.Surgical Site Infection, Mediastinitis, Following Coronary Artery Bypass Graft (CABG):.Catheter-Associated Urinary Tract Infection (UTI).Secondary Diabetes with Hyperosmolarity.Manifestations of Poor Glycemic Control.For FY 2014 through FY 2023, there are no additional HAC categories added: 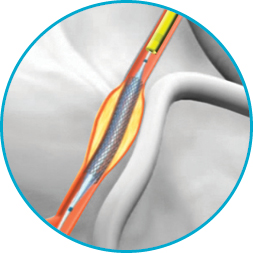 
These 14 categories of HACs listed below include the HACs from the IPPS FY 2013 Final Rule which are Surgical Site Infection Following Cardiac Implantable Electronic Device (CIED) and Iatrogenic Pneumothorax with Venous Catheterization. The IPPS FY 2009 Final Rule is available in the Statute/Regulations/Program Instructions section, accessible through the navigation menu at left. On July 31, 2008, in the Inpatient Prospective Payment System (IPPS) Fiscal Year (FY) 2009 Final Rule, CMS included 10 categories of conditions that were selected for the HAC payment provision. Payment implications began October 1, 2008, for these Hospital Acquired Conditions. Section 5001(c) of Deficit Reduction Act of 2005 requires the Secretary to identify conditions that are: (a) high cost or high volume or both, (b) result in the assignment of a case to a DRG that has a higher payment when present as a secondary diagnosis, and (c) could reasonably have been prevented through the application of evidence‑based guidelines. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
